For years, semaglutide existed as a paradox, especially in India. Although the drug is clinically proven and continues to be in high demand, it remained inaccessible to many patients. That paradox is starting to change, with the focus shifting from whether the drug works to how quickly and widely it can be accessed after the semaglutide patent expiry.
Semaglutide Patent Expiry Timeline: The Inflection Point
Semaglutide’s journey in India has been shaped more by exclusivity than by science. Till now, the drug, also known as a leading GLP-1 receptor agonist, has been developed & marketed exclusively by Novo Nordisk. Access in India remained tightly limited under patent protection. That changed on March 20, 2026.
This date represents more than the end of a patent term. It marks the transition of the drug from a patented innovation to an open-market opportunity. Historically, these transitions in India have led to rapid changes in pricing and adoption. Semaglutide is set to follow a similar path, especially given the size of its target market.
Price Drop Potential: A 70% Reset in Access
One of the most immediate consequences of semaglutide patent expiry is pricing disruption. Early market movements already indicate a 50–60% reduction in therapy costs, with the potential to reach up to ~70% as competition deepens.
This patent expiry is transformative, particularly in the Indian context, where healthcare expenditure remains out-of-pocket for most individuals. Hence, affordability determines the rate of therapy adoption. This therapy price reduction does not just lead to incremental improvements in access, as such, it fundamentally expands the pool of patients who can realistically consider long-term treatment.
This shift is likely to:
- Improve treatment initiation rates.
- Enhance adherence to chronic therapy.
- Enable earlier intervention in metabolic conditions.
From this perspective, pricing moves from a barrier into a powerful enabler of care.
Key Players Entering: The Rise of India’s Generic Powerhouses
With the patent barrier removed, the competitive landscape is rapidly opening up. More than 50 Indian pharmaceutical companies are expecting to enter the GLP-1 market after semaglutide patent expiry. Some of the renowned players are Sun Pharmaceutical Industries, Dr. Reddy’s Laboratories, Biocon, Zydus Lifesciences, Glenmark Pharmaceuticals, Torrent Pharmaceuticals, Natco Pharma, Alkem, Mankind Pharma, Eris Lifesciences, Lupin, Cipla, Abbott India, and Emcure Pharma.
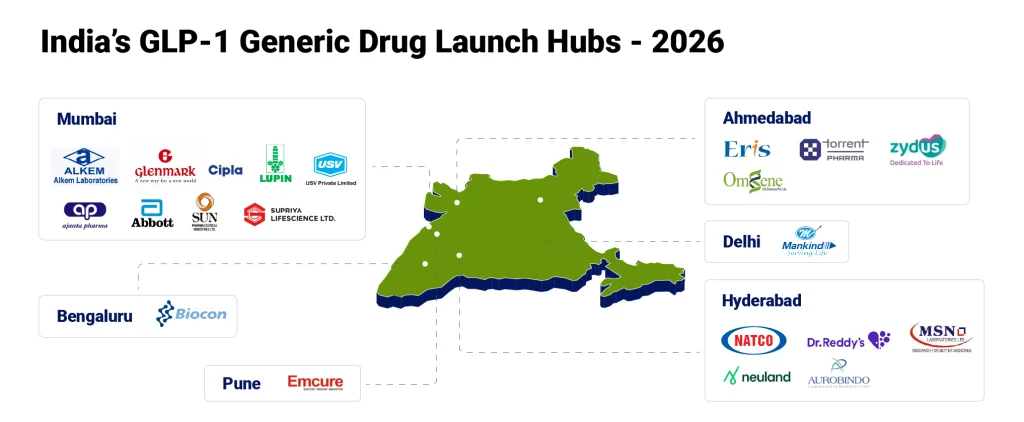
But this is not a standard generic story. A multi-player market channels competition along other dimensions beyond price. Differentiation will come from:
- Scale & consistency of manufacturing.
- Novel delivery formats.
- Physician engagement.
- Distribution beyond Tier 1 and Tier 2 cities.
Execution excellence may be a more relevant concept, shifting the competitive frontier from molecule differentiation to operational excellence.
Behind the Launch: What Companies Are Really Investing
While discussing key elements related to the launch of semaglutide, pricing, and competitiveness, this also hints at what’s behind the drug launch story. Unlike typical generics, this is a complex peptide therapy requiring investments in dedicated manufacturing and separate delivery systems.
Investments in different therapy assets, such as reusable injection pens, oral, and vial, are already evident. This reflects a visible shift from pure price competition toward skill-based execution.
With policy support like the ₹10,000 crore “Biopharma Shakti” initiative, the broader direction is clear. This is not just about entering the market, but about building long-term capability.
Markets Opening: Emerging Economies vs. Developed Strongholds
The impact of the semaglutide patent expiry is not uniform worldwide. However, this event creates a clear divide between emerging and developed markets.
The introduction of generics in countries like India, China, Brazil, and Canada is likely to unlock large, inaccessible patient populations. These markets are currently highly price sensitive, making them highly responsive to affordability-driven expansion.
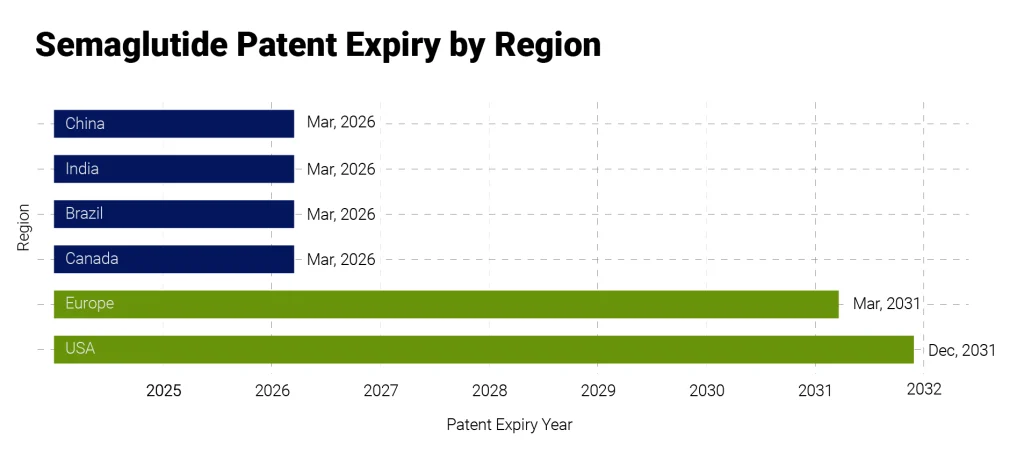
In contrast, regions such as the United States and the European Union continue to operate under patent protection for now. Semaglutide is still positioned as a patented therapy there, and its growth is majorly driven by innovation and lifecycle management rather than price-based adoption.
This divergence reflects a broader trend: while developed markets focus on value maximization, emerging markets are becoming the engines of volume growth.
Impact: Patient Access and Market Expansion
The most meaningful impact of this transition will be felt at the intersection of access and outcomes.
Following the semaglutide patent expiry, a meaningful impact on access and outcomes is expected. India carries the dual burden of rising diabetes and obesity, where a significant proportion of the population is either undiagnosed or inadequately treated. Hence, semaglutide’s dual action concerning glycemic control and weight management makes it a significant therapy in this regard.
As affordability improves, several shifts are likely to unfold:
- A larger share of the population becomes treatable
- Physicians can recommend therapy earlier in the disease lifecycle
- Management at this level could potentially reduce long-term complications.
With expanded access, new challenges come up at the same time. The possibility for off-label or unsupervised use of the medication, particularly for weight loss, may increase. Additionally, despite price reductions, some patients may still find it expensive due to an extended treatment course.
Consulting Perspective
Semaglutide’s patent expiry in India is not just a market event, it is a structural shift. It marks the transition of a high-impact therapy from limited access to broader availability, with the potential to reshape both treatment patterns and market dynamics.
The next phase will not be defined by the molecule itself, but by how effectively the broader healthcare ecosystem; including manufacturers, providers, and patients; adapts to this new reality.
Because in the end, the real measure of success will not be how much the market grows, but how many more patients it is finally able to reach.