Research Overview
Researchers at Oregon State University’s Faculty of Pharmaceutical Sciences suggest that vaccines that stimulate the production of proteins essential to the skin’s antioxidant network may help patients strengthen their defences against skin cancer.
Messenger RNA vaccines such as the COVID-19 candidate by Moderna and Pfizer can stimulate the production of the protein TR1 in skin cells and reduce the risk of UV-induced cancer and other skin problems.
TR1 is an abbreviation for thioredoxin reductase. Reductase is an enzyme that promotes a reduction reaction in which a chemical species acquires electrons. It is usually part of a “redox” reaction in which another species oxidizes or loses electrons. The TR1 is a crucial component of the melanocyte antioxidant system. Antioxidants remove electrons from molecules inside cells and protect reactive oxygen species (ROS) that damage DNA.
Research Focus
Skin cancer is the most frequent type of cancer in the United States, according to the Centres for Disease Control and Prevention. Melanoma, the deadliest kind of skin cancer, is a type of malignant cell that accumulates inside skin cells called melanocytes. Melanocytes generate the pigment melanin, which is responsible for the skin’s colour.
A study by Arup and his colleagues using a mouse model to study the role of TR1 in skin cell health and stability was published in the Journal of Investigative Dermatology.
People at high risk of skin cancer and working outdoors in sunny climates can ideally be vaccinated once a year. Despite the presence of other antioxidant proteins, researchers have observed increased oxidative stress and DNA damage without TR1, so a TR1-only vaccine without other antioxidants may be sufficient. However, other antioxidants such as glutathione peroxidase and superoxide dismutase may also be necessary. So, everything needs to be tested and validated in the preclinical model.
Impact on Patient (End-Users)
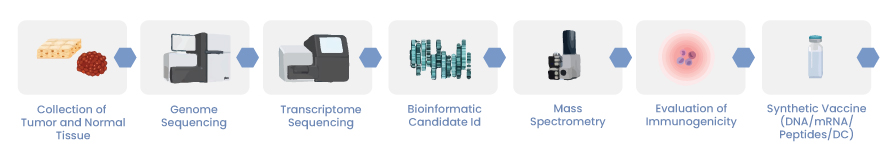
A method of strengthening the protection of a body comprising cancer including cancer by adjusting a physical antioxidant comprising cancer by preparing a localized mRNA vaccine and regulating a physical antioxidant comprising cancer Methods of monitoring are mentioned.
There are different cancer vaccine platforms being utilized by different pharmaceutical companies for example neoantigen, antigenic essence, cellular, tumour-associated antigens, and oncolytic viruses.
Neoantigens include antigens produced by tumour viruses integrated into the genome and antigens originated by mutant proteins, which are abundantly expressed specifically in cancer cells and have strong immunogenicity and tumour heterogeneity.
There are no yet approved neoantigen vaccines, but according to a ClinicalTrials.gov search, there are 160 cases of neoantigen studies and none of them entered the third clinical phase (not considering HPV vaccines). Many of these studies have shown the good application value of neoantigen.
Potential Advancements for Peer Markets
It is necessary to create an mRNA vaccine, deliver it locally or systematically, and then monitor how it enhances the body’s defences. Of course, we are at the tip of the iceberg. However, the potential to prevent the progression of various types of diseases, including cancer, by regulating the body’s antioxidant system is exciting.
Research on mRNA vaccines appears to be promising for other uses, such as cancer treatment, but these approaches have not yet been proven. Early insights from ongoing clinical trials of the mRNA cancer vaccine allowed researchers to quickly learn more about the safety and efficacy of these treatments.
Shifting Industry Trends
The market for cancer vaccines is segmented by vaccination type, vaccine use, end-user, and geography. Antigen vaccines, anti-idiotypic vaccines, dendritic cell vaccines, whole-cell vaccines, and DNA vaccines are the several vaccine kinds available on the market. Prophylactic (or prophylactic) vaccines and therapeutic vaccines are the two types of vaccines available on the market. The HPV and hepatitis B vaccines are the only preventative vaccines approved in the US. Autologous cancer vaccines (produced by yourself) and allogeneic cancer vaccines are the two types of therapeutic vaccines (made from non-cancer cells in your body).




