In today’s world, there is no scarier disease than cancer. Cancer is often considered deadly and untreatable, supported by the fact that cancer has been one of the crucial causes of mortality globally. In 2020, there were approximately 19.3 million new cancer cases and almost 10.0 million cancer deaths. However, now with Medicine, Biotechnology, and the Pharmaceutical industry advancing at such a good pace, it is safe to say that not all cancers are deadly and untreatable. It is possible to get rid of cancer if diagnosed at the right time with various new and emerging Immunotherapies. Immunotherapy is a treatment technique for cancer that aids your immune system in fighting cancerous cells. There are multiple types of Immunotherapies used for cancer treatment, such as:
- Immune checkpoint inhibitors: This is a treatment involving drugs that can block the immune checkpoints, thereby allowing the immune systems to function fully activated and more potent than normal.
- Monoclonal antibodies: Monoclonal antibodies can be designed according to the needs and administered into the immune system to fight cancer.
- Treatment Vaccines: It is designed to boost immunity and function with an enhanced ability to fight cancer. Differs from normal preventive vaccines as it is administered to patients who already have cancer.
- Immunomodulators: Drugs that boost immune parts of the immune system to treat cancer.
- Cytokines and oncolytic viruses: This treatment incorporates cytokines and modified viruses to boost immune responses against cancer.
- T-cell transfer therapy: A method that involves selecting immune cells from a patient’s tumor and modifying them to kill the tumor and then incorporating them back into the patient, thus fighting cancer. CAR T-cell therapy is one such emerging treatment for cancer.
What Is CAR T-Cell Therapy?
CAR T-cell therapy is a gene therapy that involves genetic modification of a patient’s T cell to create an immune response and fight against cancer. The treatment employs modified T cells known as chimeric antigen receptor (CAR) T-cells to recognize and kill cancer cells. T cells are taken from the patient and genetically engineered. A chimeric antigen receptor (CAR) is transmitted into the cells via a viral vector, allowing T cells to recognize better and destroy cancer cells. Antigen receptor cells multiply and destroy target cells as they realize the patient’s cells’ desired surface structure.
How Is It Done?
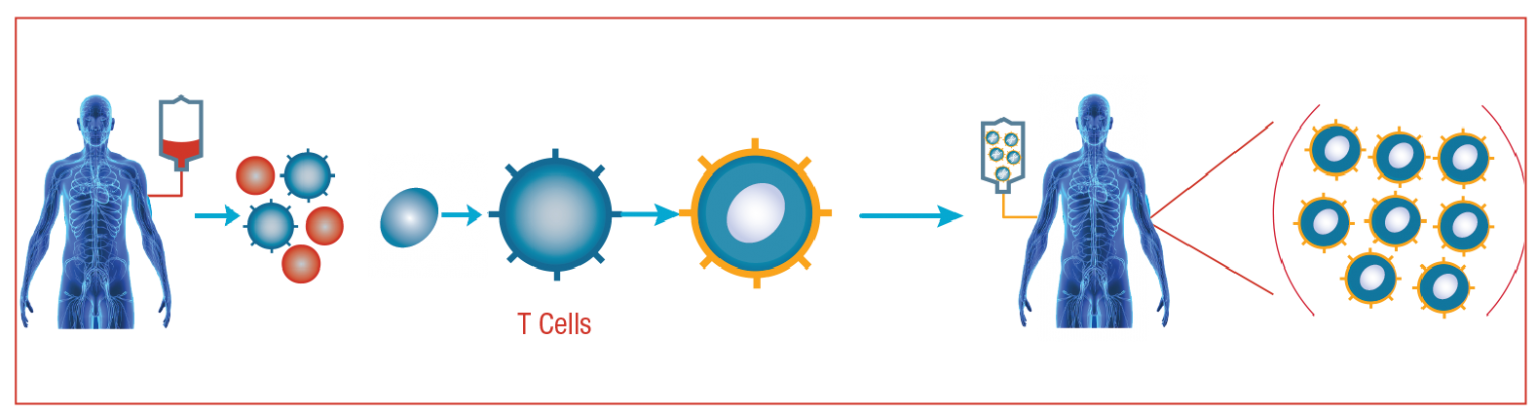
CAR T-cell therapy will take several weeks to complete. In general, it entails:
1. T cells are extracted through leukapheresis, which is a non-invasive blood removal technique.
2. Boosting T cells: Scientists position a protein called chimeric antigen receptor (CAR) on T cells’ surface to serve as an antibody.
3. Cell growth and harvesting: The cells are multiplied in the millions and frozen.
4. Infusion: While waiting for the boosted CAR T cells to return, patients receive chemotherapy. When the cells re-enter a person’s body, they begin to grow.
5. Monitoring & Treatment: Patients are monitored and treated in the hospital for two to three weeks after receiving the infusion. Fever, nausea, headache, rash, rapid heartbeat, low blood pressure, and difficulty breathing are all cytokine release syndrome signs, a severe and common side effect. As a result, CAR T care in an inpatient environment necessitates a higher degree of supervision.
CAR T-Cell Therapy Side Effects
This treatment is considered efficient and effective for the treatment of cancer. However, it has also been observed to exhibit some serious side effects, so the need for advancement in this therapy exists. CAR T-cell therapy’s side effects, especially a disorder known as cytokine release syndrome (CRS), can be fatal. Patients undergoing CAR T-cell therapy can be inundated with cytokines, which attack healthy cells and tissue, causing flu-like symptoms, high fevers, life-threatening neurological side effects, and other severe complications. In some patients, CAR T-cells can attack healthy B-cells, raising the risk of infection. Also, CAR T-cell therapy has several drawbacks, such as its limited availability to most patients, approvals being limited, and it often causes severe side effects in some patients. It can also affect the brain, causing confusion, speech problems, and even seizures. The first person to undergo CAR T-cell therapy did so less than a decade ago. As a result, physicians are still unaware of any long-term dangers.
CAR T-Cell Immunotherapy Against Types of Cancers
CAR T-cell therapy may be successful in cases where other treatments have failed. Unlike chemo and radiation, which destroy both healthy and cancerous cells, immunotherapy is more precise in targeting tumors. It is known to treat various cancers such as colorectal cancer, colon cancer, ovarian cancer, pancreatic cancer, liver cancer, lung cancer, acute lymphoblastic leukemia, non-Hodgkin lymphoma, acute myeloid leukemia, relapsed or refractory lymphoid malignancies, hematological malignancies & multiple myeloma. In 2017, the FDA approved the first CAR T-cell therapy. For the treatment of advanced B-cell lymphomas, several CAR T cell therapy agents have been approved, such as Kymria™ (tisagenlecleucel), to treat children and young adults up to the age of 25 with B-cell precursor acute lymphoblastic leukemia (ALL). Yescarta™ (axicabtagene ciloleucel) for the treatment of adults with diffuse large B-cell lymphoma (DLBCL). In 2021 February, Breyanzi (lisocabtagene maraleucel) was approved to treat adult patients with relapsed or refractory large B-cell lymphomas. Breyanzi is a CD19-directed chimeric antigen receptor (CAR) T-cell immunotherapy.
Shortcomings and Need for Further Advances
The need for increased efficacy and performance of this therapy is one of the driving forces to continue advancements. Besides, minimizing the side effects without compromising the therapeutic effects has been a challenge. This technique is also costly and requires multi-skilled labor for successful implementations, limiting its easy availability and affordability to the patients. Thus, cost minimization of CAR T cell therapy is a prime need. High relapse rates and resistance were encountered, necessitating the development of engineered CAR-T cells that were more effective for therapeutic usage. One primary need to invent new approaches is that it’s been difficult to apply CAR T cell therapy to solid tumors because it’s difficult to target the tumor as the cancer form isn’t linked to any particular surface structure.
Recent Advances and New Approaches in CAR T-Cell Therapy
- Since one of the main concerns with this therapy was its non-applicability on solid tumors, there have been considerable research and studies to succeed. A key point was found to be modulating immune regulation within solid tumor tissue. CAR T cells were also engineered to release a transgenic cytokine in response to CAR signaling in the targeted tumor tissue to create a proinflammatory setting. The TRUCKs (“T cells redirected for antigen‐unrestricted cytokine‐initiated killing”), also called “4th generation” CAR T cells, combine the direct antitumor attack of the CAR T cell with the immune-modulating capacities of the delivered cytokine.
- Research studies have been done to discover ways to lower the chance of anti-CAR immune responses to CAR T cell products on their way to clinical development.
- In February 2021, Novartis received approval for the manufacture and supply of its CAR T-cell therapy for adult patients.
- Also, Swiss pharma announced their therapy’s success for young patients and children, which was a breakthrough.
- A recent study published found a way to apply CAR T cell therapy to solid tumors: American and Finnish researchers collaborated to develop a new way of programming CAR T cells to destroy only cancer cells while leaving healthy cells with the same marker protein as cancer cells alone.
- A new technique based on ultrasensitive HER2 cell recognition is currently being investigated to battle breast, ovarian, and abdominal cancer. The researchers developed a new CAR T cell engineering technique focused on a two-step recognition method of HER2-positive cells. The researchers were able to engineer a response in which CAR T cells destroy only the cancer cells in the cancer tissue thanks to the engineering.
- The most severe drawback of CAR T-cell therapy is cytokine release syndrome. Researchers in the United States and Italy found a way to prevent the side effects in leukemia patients who received CAR T therapy in late May 2020. Drugs to block a vital molecule in the syndrome were used in one mouse model study. The T cells in the second study, which used a mouse model, were genetically modified to prevent the side effects. This could lead to CAR T treatments that are both safer and more advanced.
- Early-stage clinical trials of “second-generation” CAR T cellular immunotherapy products have shown that they can resolve resistance, reduce side effects, and ease treatment.
- Allogeneic CAR T Cell Therapy is being developed, which has changed the game. Compared to other available CAR T cells, which involve using a patient’s own genetically modified T cells produced through a multi-week manufacturing process, they could be manufactured in advance and stored for immediate, off-the-shelf use. CAR T cell cytokine release syndrome and neurotoxicity are avoided and also are lower in cost.
- Cellular immunotherapy for B-cell cancers will one day be available over the counter. FATE and Allogene are pioneering the development of allogeneic “off-the-shelf” CAR T cell therapies. Their progress was recently updated, claiming that the time-consuming and expensive treatment method with CAR T cells is no longer needed as an off-the-shelf product emerges.
- FT596, cellular immunotherapy focused on off-the-shelf NK cells – the immune system’s “first line of protection” is also the first cellular immunotherapy to be genetically modified to produce three active antitumor components.
- CAR T cells that target one or more antigens other than CD19 specific to various cancer types are being produced and tested.
- Novel CAR-T cell-based therapies or combination therapies along with CAT-T cells may bring new hope for multiple myeloma patients. Experiments with CAR-T cells in Multiple Myeloma patients showed that they have a lot of therapeutic potential.
- Autologous CAR T-cell therapy is the personification of individualized therapy; however, large-scale CAR T-cell development is complex due to this personalized aspect.
- CliniMACS Prodigy (Miltenyi Biotec) is one such technique. It’s a closed automated system that provides integration solutions by manufacturing cells with closed-tubing pathways and delivering “CAR T-cell in a box.”
- Outpatient CAR T-cell therapy has emerged, making it safer and less toxic. Evidence suggests that outpatient stem cell transplant results in comparable or better QoL, PROs, and cost-effectiveness than inpatient stem cell transplants.
- Decentralizing the manufacture of CAR T-cell production: Decentralizing CAR T-cell manufacturing has many benefits, including lower production costs, faster time from collection to infusion, and the potential to scale rapidly without the need for new large-scale manufacturing plants.
- Development of “armored CARs” that co-express proinflammatory cytokines like IL-12 or IL-15 to boost CAR T-cell proliferation and persistence in the face of tumor-mediated immunosuppression.
- CARs that are “affinity-tuned” or engineered to have a lower affinity for the target antigen could potentially limit CAR targeting to tumor cells that overexpress the antigen significantly.
Future Potentials
- Standard measure protocol to be developed to systematically monitor and comprehend the possible effects of immunogenicity during trials involving CAR T cells and routine clinical practices.
- Streamlining the manufacturing processes of CAR T-cells.
- CAR T-cell therapies are being tested for ways to enhance outcomes, reduce side effects, expand the target beyond CD19, and target solid tumors.
- Automated CART production.
- AI for labor and production services.
T-cells are programmed to recall harmful cells or viruses that they destroy in the future. CAR T-cells are no exception. If the procedure is followed correctly, they can be on call 24 hours a day, seven days a week for the rest of the patient’s life. If cancer cells reappear, the CAR T-cells should recognize them and kill them, even if the patient is unaware of it. CAR-T cells are made to have long-term effects after only one administration. In general, once you inject the engineered CAR T-cells into your body, they stay there. It is equivalent to keeping a “living drug” in your system for the rest of your life.