Strategic Development
Rarecells Inc., a leading Liquid Biopsy company, announces the development of a revolutionary and accurately targeted digital single-cell Next-Generation Sequencing (scNGS) for single-nucleotide variant (SNV) method to detect mutant genes of pancreatic ductal adenocarcinoma (PDAC) circulating tumor cells (CTC) enriched by the ISET technology.
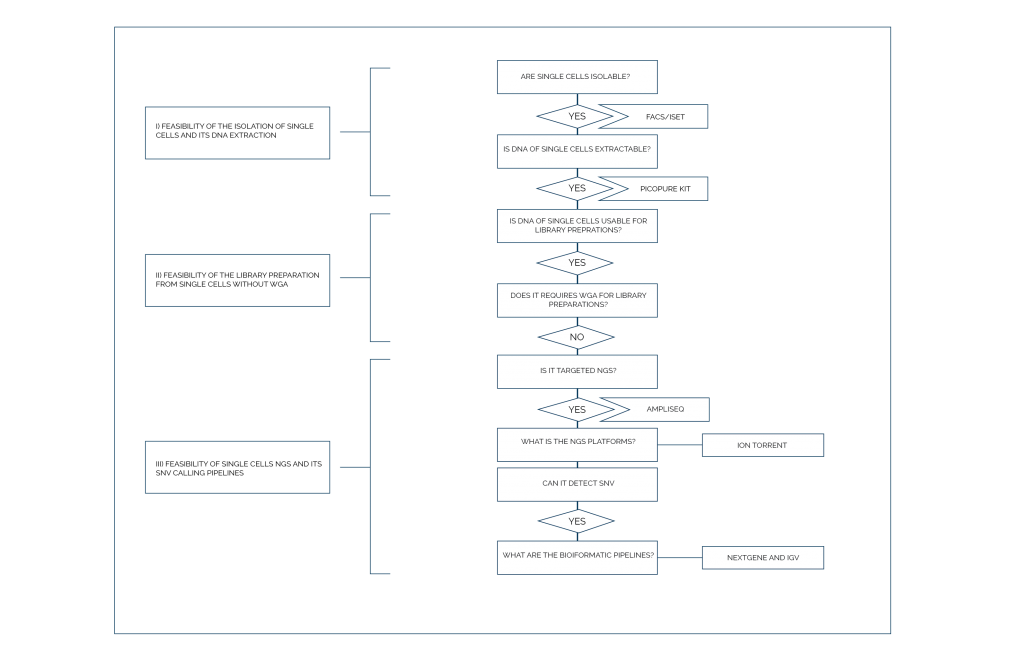
Strategic Alignment
Using targeted single-cell next-generation sequencing (scNGS), a new method for SNV analysis without preamplification has been developed. This approach is based on digital next-generation sequencing (dNGS). It uses 45 g. e. (150 pg) of cell-free DNA (cfDNA) and can detect low-abundance cfDNA mutations (0.1% to 1%) in pancreatic juice samples collected from the duodenum of pancreatic cancer patients. The company’s Strategic focus is on the ambition to detect pancreatic ductal adenocarcinoma (PDAC) before the metastasis stage. The procedure for identifying and classifying circulating malignant cells is less than one-tenth of the total cost compared to single-cell NGS methods.
Immediate Impact
The innovation for SNV analysis without preamplification using targeted single-cell next-generation sequencing (scNGS) is highly cost-efficient. The method uses only one-tenth of the cost compared to other single-cell NGS methods.
The detection of pancreatic cancer before the development of metastases and treating patients when these circulating cancer cells are still in the bloodstream but have not yet developed metastases may have a greater chance of beating cancer. This latest approach is an accurate way to diagnose cancer and its recurrence and a monitor marker for surgical or chemotherapeutic treatments.
Budding Growth Opportunities for End Users
This new sequencing technology can be used in a wide range of applications, including custom-designed panels with unique gene interests in any malignancy. It can improve the diagnostic accuracy of cytopathology specimens in low-cellularity biopsy samples. It may also be used to investigate intra–tumoral heterogeneity in the primary tumor.
PDAC is a lethal disease with a survival rate of less than 10% after five years. Surgical resection of the local disease offers the best chance of cure, but about 80% of resected patients die, with a systemic recurrence pattern being the most common. As a result, a better understanding of systemic disease mechanisms after margin-free resections will help in the production of more effective therapeutic strategies.
Potential Advancements for Peer Markets
The novel method of detecting CTCs holds significant promise in the general understanding of all types of cancers, and thus holds great promise across the entire spectrum of the disease, from screening to early detection, clinical decision-making, and treatment selection, as well as in assessing treatment response, prognosis, and eventual survival outcomes.
Across the Globe, about 18.1 million new cancers were diagnosed, and 9.6 million people died of the disease. Therefore, a novel method of detecting CTCs is an effective, non-invasive way to diagnose cancers earlier and has the capability to save millions of lives each year.
Shifting Industry Trends
According to previous studies using ISET Technology, the presence of a mesenchymal-like CTC phenotype that expresses both epithelial and mesenchymal markers has been linked to cancer recurrence in patients with pancreatic cancer.
CTC research is advancing at a breakneck pace, with new technology emerging to keep pace with a continuous maturing understanding of CTC sources, forms, and properties. New and updated technologies are important for improving our understanding of CTCs and obtaining the critical clinical insight needed to help cancer management and care.
In this novel method, the ISET technology was used to gain access to this unusual CTC phenotype for susceptible and specific targeted detection of mutated PDAC CTCs using a novel WGA-free digital scNGS workflow. This new sequencing technology has the potential for a wide range of applications, including the use of custom-designed panels with unique gene interests in any malignancy.
In comparison to the existing and widely used immunofluorescent procedure as a liquid biopsy biomarker for cancer detection, the technique can be used to identify the genetic features of CTCs from the peripheral blood of patients with PDAC, allowing for more precise identification of CTCs.