T-cell engagers (TCEs) are unlocking an innovative oncology segment by bridging cytotoxic T-cells directly to tumor cells. In the TCEs, specifically, the CD3-binding portion serves as the mechanism of action. While multiple scientific studies have centered on tumor-antigen selection and multispecific formats, it highlights the CD3-binding domain as the mechanistic core defining how next-gen therapies are differentiated.
Why Does CD3 Binding Determine the Therapeutic Window in TCEs?
The CD3-binding domain binds with T-cells to form the CD3 complex, which activates T-cell and cytotoxic effector responses. But here is a catch. Activation levels of T-cells play a critical role, as insufficient activation makes the cancer therapy ineffective. On the other hand, excessive or poorly tuned signalling can drive cytokine release syndrome (CRS) and systemic toxicity.
Therefore, the CD3 binding domain is considered a primary determinant of the therapeutic window for T-cell engagers. Consequently, companies are no longer claiming generic “CD3 binding domain” sequences. Instead, they are seeking specific binding epitopes, structural features, and quantitative affinity/avidity ranges that determine how, when, and where CD3 is engaged.
How CD3 Binding Innovation Is Reshaping the Patent Landscape?
Advancements in T-cell engager design have significantly redefined the IP landscape for CD3 binding domains, gradually shifting from general claims to highly specialized protections surrounding sequences, epitopes, and function. Additionally, differentiation is no longer determined solely by target selection but by precise control of T-cell engagement, which comprises optimizing binding affinity, enhancing specificity to reduce off-target effects, and improving the overall therapeutic efficacy of the T-cell engagers.
As a result, innovation is increasingly defined by how CD3 is bound, activated, and converted into clinical effectiveness.
Phase I: Broad Platform Claims Laid the Foundation
The first wave of T-cell engagers (TCE) patent families/ early TCE patent families, particularly those related to BiTE (bispecific T-cell engagers) platforms, established broad-based architecture-level claims. Many filings described bispecific constructs containing a CD3-binding domain (typically directed towards CD3ε) combined with a tumor-associated antigen (TAA) binding domain.
For instance, Amgen’s early filings (WO2017134158) describe how they link a CD3-binding domain to a tumor-targeting arm in bispecific molecules. Most early-filed patents lacked distinction amongst specific CD3-binding sequences or finely tuned functional attributes due to their purpose being to provide the widest possible claim scope. Subsequent innovations, optimizations, and competitive claim space for CD3-binding epitope selection and biophysical properties followed.
Phase II: Precision Engineering of the CD3 Binding Arm
As the TCE field has expanded and diversified, claim strategies have moved from broad conceptual frameworks to precision-engineering of the CD3-binding arm at the sequence and epitope levels. Recent patent families describe CD3 binders based on the following:
- Specific antibody variable region and CDR sequences.
- Discrete epitopes on CD3 epsilon or CD3 heterodimeric subunits.
- Tightly controlled binding affinities.
- Functional endpoints (e.g., cytokine attenuation while preserving cytotoxic potency).
For example, newly filed applications (e.g., US20230075633 and accompanying filings) disclose engineered anti-CD3 antibodies with enhanced biophysical stability, developability, and safety (including decreased risk of CRS and improved pharmacokinetics). Collectively, this reflects a transition from platform-level IP rights to molecule and epitope-level protection; minor changes in sequence, epitope specificity, or affinity profiles may serve as grounds to establish separate patent eligibility and distinct therapeutic profiles.
Affinity Tuning: Engineering Safety into the Claims
A significant advancement in the design of the CD3-binding arm has been deliberate affinity modulation. The first-generation TCEs included high-affinity CD3 binders, which delivered strong T-cell activation. However, this improvement was accompanied by a higher likelihood of adverse effects, including cytokine release syndrome.
To broaden the therapeutic window, several groups have subsequently developed attenuated (lower-affinity) CD3 binders, engineered to dampen systemic T-cell activation and/or cytokine release while retaining potent and tumor-selective cytotoxicity. More recently filed patent applications, therefore, often include claims directed to defined CD3 affinity windows or binding kinetic parameters, explicitly linking these profiles to improved safety and pharmacodynamic behavior.
For example, disclosures such as WO2015006749 outline CD3-binding domains that balance efficacy with reduced adverse effects. Thus, rather than focusing on structural definitions alone, functional characteristics, such as affinity, kinetics, and cytokine modulation, represent an increasingly important aspect of patentability in the CD3-binding TCE space.
Epitope-Level Claims: Owning Where CD3 Is Engaged
An additional dimension in CD3-related intellectual property is epitope specificity. CD3-binding antibodies usually do not always recognize and bind to the same region of the CD3 complex; variations in epitope-specific recognition can significantly affect both signal intensity/ pathway utilization and overall safety profiles.
Therefore, companies are progressively drafting claims directed to antibodies binding to particular epitopes on the CD3 complex, often supported by structural, mutagenetic, or epitope-mapping data. This leads to differentiation of their products from prior-art CD3 binders. Under this scenario, a layered IP landscape comes into play, in which one company may own a particular epitope(s), another may file for a distinct epitope(s)/binding manner(s), and a third company may emphasize functional aspects (e.g., cytokine suppression or activation thresholds) relevant to its epitope-specific binder.
At this granular level of detail, design-around strategies for previously filed CD3-related patents usually require considerable mapping efforts regarding epitopes, binding modes, and functional readouts to mitigate both direct and doctrine-of-equivalents infringement.
Format as a Differentiator: CD3 Binding in Structural Context
The CD3-binding domain operates within a well-defined molecular architecture; modern patent claims generally reflect this interaction by defining specific connections between CD3-binding properties and molecular formats, which include:
- BiTE‑type bispecific antibodies lacking an Fc region.
- IgG‑like bispecific antibodies that retain an Fc domain.
- Trispecific or multispecific constructs incorporating additional antigen‑binding arms.
To illustrate, patents, such as WO2017134134, specify CD3-binding domains integrated into well-defined scaffold configurations, where the selected format affects other crucial attributes like stability, serum half-life, and Fc-mediated immune engagement. Therefore, even if a prior-art disclosure describes a specific CD3-binding sequence, its integration into a specific molecular configuration, including a defined domain arrangement, linker design, and valency, can support patentable subject matter.
Patent Landscape and Key Players in T-Cell Engager Innovation
If you look at how T-cell engager technologies have evolved over the past decade, the shift is pretty clear. What used to sit on the fringes of research has steadily moved into the spotlight, becoming one of the more active areas in next-generation immunotherapy.
Back between 2010 and 2018, there wasn’t a huge amount happening on the patent front. Filings stayed relatively low, usually under 30 patent families a year. But around 2019, things started to change. Things really started to pick up after that. Filings moved from 46 in 2019 to 178 in 2025, which is a pretty steep jump in just a few years. That kind of rise usually means the space is gaining real traction. Not just curiosity anymore, but stronger belief in the science, more money flowing in, and a lot more activity around testing these therapies, especially in cancer and immune-related areas. As for 2026, the lower number doesn’t really suggest a slowdown; it’s more likely just because the year hasn’t played out fully yet.
Patent filing patterns tend to move with how research priorities shift over time. The priorities for innovation change as new technical challenges or new opportunities arise. Regulatory requirements, particularly evolving environmental standards, frequently drive the setting of priorities and the pace of particular innovations. At the same time, companies are adopting a more open and collaborative business approach, actively sharing ideas and co-developing solutions. This shift is reshaping how they capture innovation and reflect it in traditional patent filings over time.
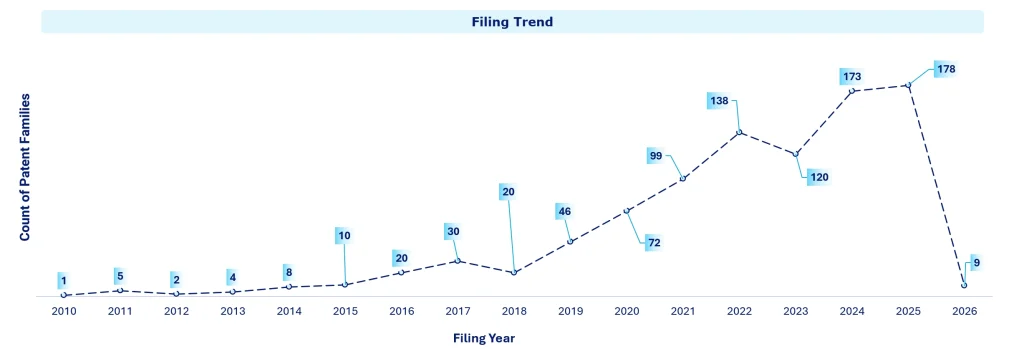
Figure 1: T-cell Engager Innovation Filing Trend
Industry Giants and Academic Players in T-Cell Engagers Patent Landscape
When it comes to who’s actually driving this space, it’s a mix rather than a single group. Big pharma and smaller biotech companies are both playing their part, just in different ways.
Amgen, for example, has clearly been in early and stayed consistent, with 51 patent families under its belt—showing long-term commitment to this area. Alongside that, companies like Marengo Therapeutics, Regeneron, Hangzhou DAC Biotech, and UTC Therapeutics are all exploring their own directions, which is really broadening how these therapies are being developed and refined.
It’s not just industry doing the heavy lifting either. Academic and research institutions are playing a big part in moving things forward. The University of Texas leads among academic patent holders, with others like Dana-Farber Cancer Institute, National Research Council Canada, Baylor College of Medicine, and California Institute of Technology close behind. Their involvement shows how important it is to bridge early-stage research with real-world application. This transition usually happens through close collaboration with industry.
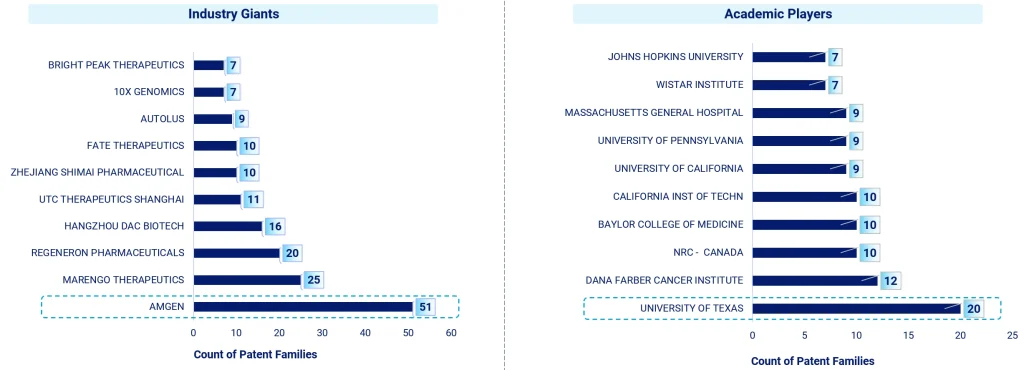
Figure 2: Industry Giants and Academic Players in T-cell Patent Landscape
Put it all together, and you get a space that’s clearly still growing and taking shape. There’s more competition now, more research activity, and a broader mix of players than before. As teams advance clinical programs and explore new targets, T-cell engagers are likely to remain a key area to watch within immuno-oncology.
Competitive Landscape of Drug Approvals by Company
Approvals for bispecific and multispecific antibody therapies evolved across different companies over time, giving a clear view of how this space grew from a single early entry into a much more active and competitive field.
In 2014, Amgen secured approval for Blinatumomab (Blincyto, CD19-targeting), making it one of the first drugs in this class to reach the market. Following this, the period went “quiet” for a while, and the development of bispecific antibodies slowly continued to advance towards building clinical confidence.
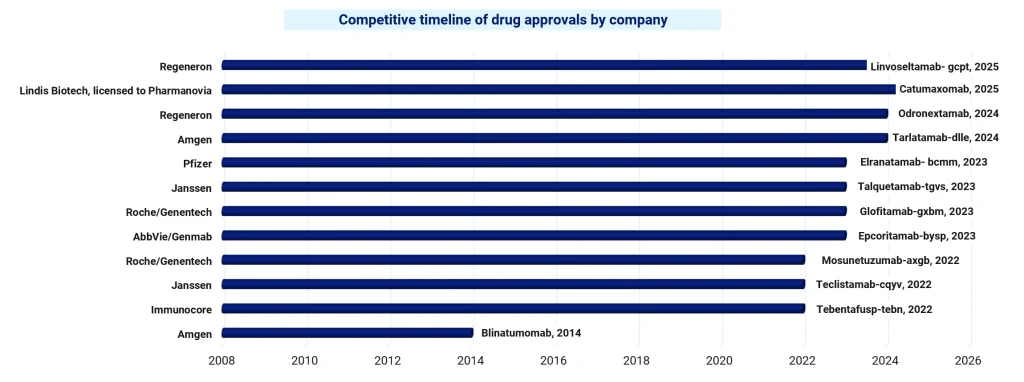
Figure 3: Competitive Timeline for Drug Approvals by Company (Source)
As we entered 2022, the number of approvals started increasing significantly. Companies like Immunocore, Janssen, and Roche/Genentech started appearing more consistently, especially around prominent targets like CD20 and BCMA. At this stage, the landscape started to feel much more active, with more players entering the competition.
By 2023, companies like AbbVie/Genmab, Roche/Genentech, Janssen, and Pfizer were all securing approvals around the same time. It was no longer just a story of a few early leaders driving the space, but multiple players moving forward together.
In 2024 and 2025, the scope broadened even further. New targets like DLL3 and EpCAM started to emerge, indicating that innovation was moving beyond the earlier focus areas. At the same time, established players like Amgen and Regeneron stayed consistently active, and partnerships such as Lindis Biotech with Pharmanovia showed how collaboration was becoming an important part of pushing these therapies forward.
From Molecule Design to Market Access: Why IP Strategy Is Non-Negotiable
Designing a new T-cell engager (TCE) involves not just science, but also strategic navigation of intellectual property considerations. Developers must deliberately consider whether their proposed CD3 binder infringes existing claims based on sequence identity; whether its binding characteristics (e.g., affinity, kinetics, and epitope specificity) fall within defined claim boundaries; and whether the chosen molecular format, such as BiTE, IgG-like, or trispecific, induces further patent coverage.
The Takeaway: CD3 Is Where Science Meets Strategy
Although TCE development usually considers tumor‑targeting specificity and multispecific architectures, the CD3‑binding domain remains the core driver and key constraint of innovation in this class of molecules.
Similarly, this trend demonstrates how precision immunotherapies are influencing the direction of developments in the IP community. In these therapies, fine-tuning activation is just as important as selecting targets. Ultimately, how the future of TCEs evolves will not solely depend on the laboratory experiments but is being shaped by decisions made in patent offices, where the competitive battle for ownership of the CD3-binding arm continues to evolve.
Building a T-cell engager? … Make sure your innovation is not boxed in by existing CD3 IP.
Assess your freedom to operate searches, uncover white-space opportunities, and build a CD3 strategy that stands up scientifically and commercially.
To understand feasible IP strategies tailored to business requirements, connect with our IP and tech experts by filling out the form below.