Overview
Chimeric antigen receptor (CAR)-T cells are genetically engineered cells primarily known for targeting and treating liquid cancers, i.e., leukemia and lymphoma. The success rate in these hematological malignancies has led to FDA approval of 5 drugs along with numerous undergoing clinical trials. Despite these positive outcomes, CAR-T for solid tumors hasn’t been quite successful. However, recent research activities have remarkably contributed to identifying the ideal target antigens and relevant “CAR” modifications to overcome repressions. As a result, even solid tumors are also being targeted effectively by CAR-T cells. The momentum gained so far has resulted in the treatment of not only cancers but also other auto-immune diseases.
Although several diagnostic and treatment options are available to detect malignant cells, control disease progression at early stages by eradicating tumor cells, and prevent remission. In this context, immunotherapies—a relatively modern inclusion in the array of anti-cancer approaches—have showcased substantial potential. Amidst the current initiatives undertaken to develop more effective targeted anti-cancer therapies, T-cell therapies [specifically CAR-T therapies and tumor-infiltrating lymphocytes (TIL) therapies] have emerged as promising options, owing to their ability to selectively target and destroy tumor cells residing in the body with minimally impacting the healthy tissues.
Current market dynamics in CAR-T cell therapy
The CAR-T cell therapy market is set to surpass $3.1 Billion in 2024 and is expected to grow at a phenomenal compound annual growth rate (CAGR) of over 20% by 2028. Cancer is among the leading causes of fatalities worldwide. Currently, more than 250 companies are engaged in developing more than 1,200 early and late-stage T-cell therapies worldwide. With the potential approval & commercialization of new therapies, expanding applications, and ongoing research and development efforts, the market is projected to grow at a substantial pace in the upcoming years.
A brief on most targeted cancers
Cancer accounted for nearly 10 million deaths in 2020 due to the detrimental impacts of the pandemic.
Exhibit 1: The most common cancer types (in terms of new cases)
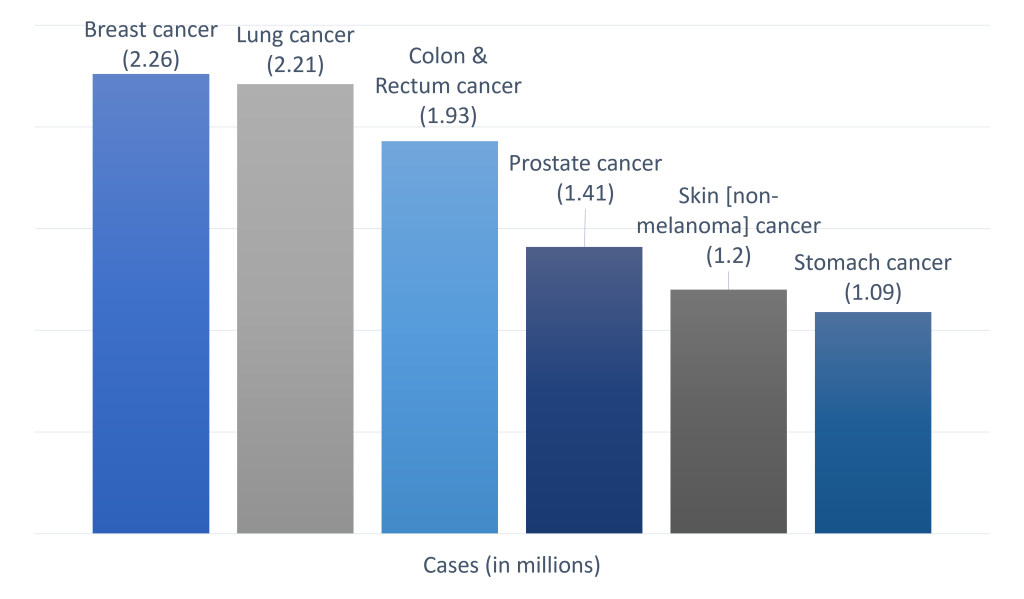
Apart from these, pancreatic, esophageal, liver, and cervical cancer are relatively less common.
How are solid tumors linked to targeted antigens?
Targeted antigens in the context of solid tumors refer to specific molecules or proteins present on the tumor cells’ surface that the immune system can recognize. These antigens can serve as targets for various therapeutic approaches, including targeted therapies and immunotherapies.
Exhibit 2: Targeted antigens for different tumor types
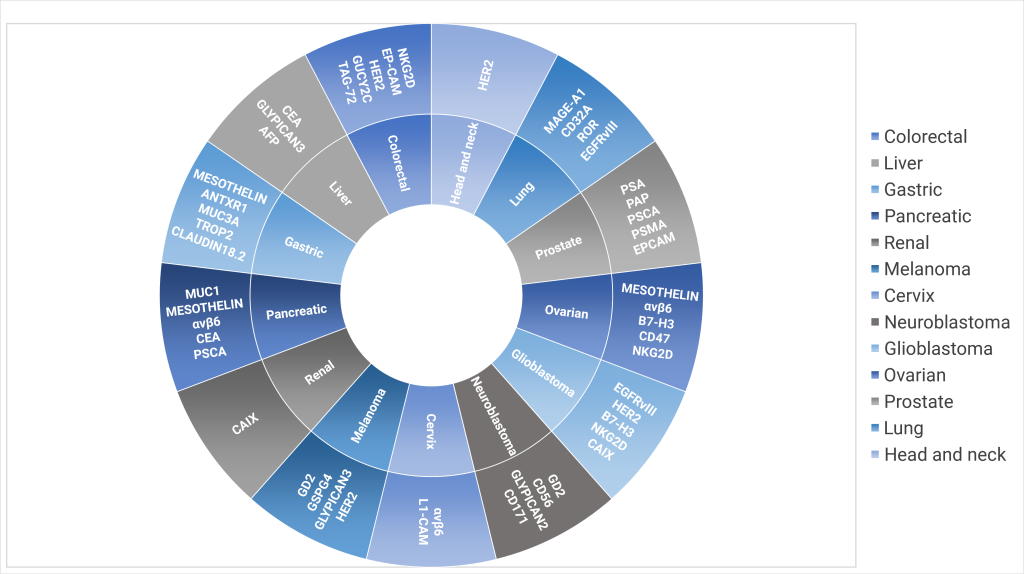
IeB’s insights into mapping cancer variants with specific targeted antigens:
- Based on the numerous recent scientific publications, besides exploring diverse antigenic targets for targeting the same cancers, efforts are being made to identify one antigen for precisely targeting multiple tumors.
- MUC3A, HER2, GLYPICAN, and MESOTHELIN are common antigenic targets.
Obstacles in targeting solid tumors through T cells
- Compared to hematological tumors, finding an ideal target antigen for solid tumors is relatively difficult. Also, the latter type rarely expresses a single tumor-specific antigen.
- Another hindrance is infiltration by T cells, as the presence of blood vessels or high endothelial venules (HEVs) is critical for T cell infiltration and has been associated with tumor regression in cancers such as melanoma.
- Survival in T.M.E is another challenge as it is hostile to them. In an inflammatory environment, tumor cells often upregulate ligands such as programmed cell death ligand 1 (PD-L1) and Galectin9, which bind to T-cell inhibitory receptors.
- Lastly, T cell activation against tumor antigens may lead to cytokine release syndrome (CRS) and neurotoxicity, which can result in severe and potentially life-threatening adverse effects.
Despite all these challenges, several players are actively developing strategies to target these solid tumors.
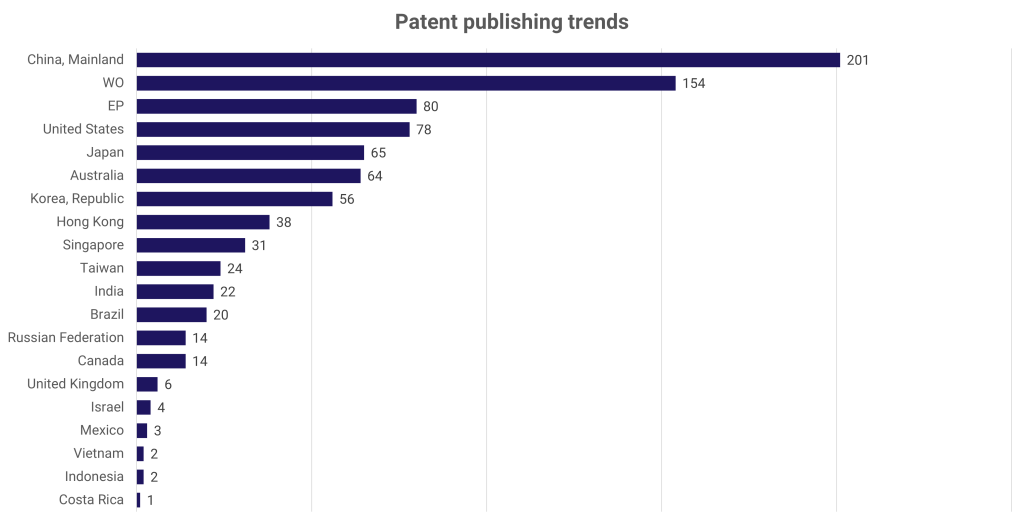
Impact of COVID-19 on cancer care-related patent filings:
The global impact of the COVID-19 pandemic has undeniably heightened public awareness regarding health and well-being. Owing to this, a significant surge in R&D efforts dedicated to advancing cancer care has been witnessed. Thus, biopharma companies and research organizations focus on parallelly accelerating innovations targeting cancer and commercializing those novel technologies.
Exhibit 3: Patent filings from 2021
**Source: IeB analysis
Exhibit 4: Global clinical trials reporting scenario
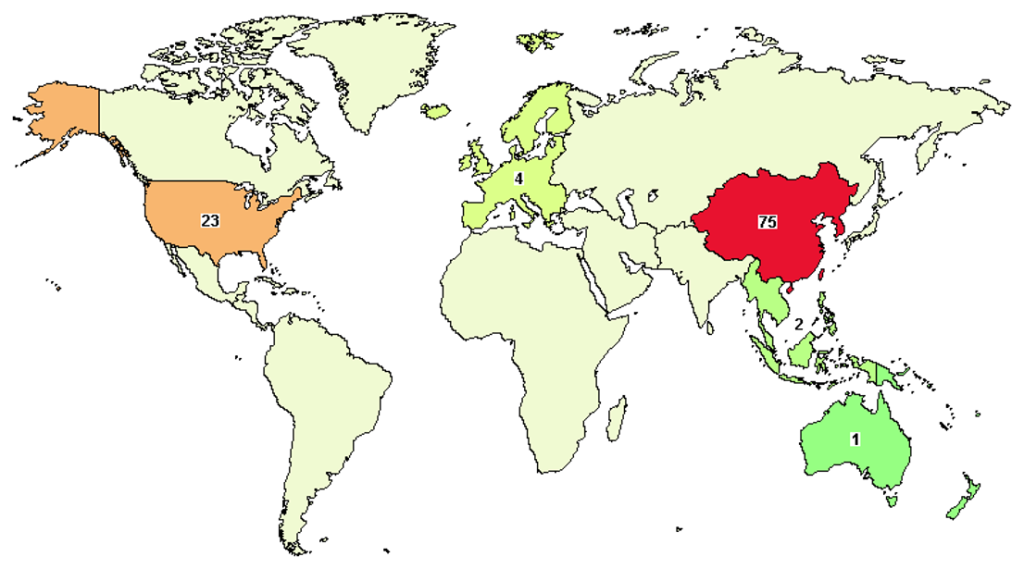
Insights:
- There are currently 125* clinical studies reported so far, with 14 trials in Early Phase 1; 97 in Phase 1; and 36 in Phase 2
- No reported clinical trials have entered phases 3 and 4
- China leads with 75 out of 114* global clinical trials being reported
Conclusion:
At Ingenious e-Brain (IeB), we stand at the forefront of technological advancement, offering invaluable technological scouting and benchmarking services that enable clients to select optimal platforms for scaling up their endeavors. Moreover, as part of IeB’s exclusive IP solutions spectrum, we extend our support by identifying available licensing opportunities, empowering clients with a keen understanding of the current landscape and potential collaborations in cancer care. We offer tailored solutions that are finely tuned to meet the unique needs of each client, ensuring a strategic advantage in navigating the intricate terrain of innovation and discovery.
With a commitment to innovation and precision, IeB accompanies as a trusted partner, empowering clients to make informed decisions and seize opportunities in the dynamic world of technology and research.
Stay tuned to dive into the wisdom of CAR-T cell therapies that are in the emerging stage, on the verge of commercialization, key players involved in such innovations, recent advancements, and a lot more!
Want to talk to our seasoned IP experts in the life science domain? Either fill out the form below or email them to contact@iebrain.com.
About Ingenious e-Brain: –
Ingenious e-Brain provides high-quality, customized, and cost-effective Technology Research, Business Research, and Intellectual Property Research solutions to industry leaders, and innovative companies across the globe. Innovation, knowledge, and transparency form the basis of our company’s mission and vision. Along with cost benefits, we provide highest quality results ensuring fool-proof confidentiality and security. We are an ISO certified company with offices in India and USA.
Ingenious e-Brain has a strong team of analysts, and subject matter experts with domain proficiency which is devoted to help clients grow. Our highly qualified professionals offer tailored, value-added and cost-effective services to our clients. We believe in building long term relationships with our clients who include national and international corporations, Fortune 500 companies, world’s leading research institutes and universities as well as independent inventors.
Get in Touch:-
USA Office:
646 Sutton Way#1006
Grass Valley, CA 95945 United States
+1-347-480-2054
India Office
207-208 Welldone TechPark, Sohna Road
Sector 48, Gurugram, Haryana 122018, India
+91 124 429 4218