Symbiotic Relation Between Innovative & Generic Pharma
Innovation in pharma indicates novel drug or drug-related discovery, thereby applying for exclusive rights for the hard work invested during R&D, and clinical trials by the innovator company to discover the blockbuster drug. This exclusivity can be acquired by various means one is applying for the patent.
But anyhow, in the end, the blockbuster drug patents also must expire one day, which will attract generic competition. This patent expiration is making the market more attractive to established generic drug makers and inviting innovators’ interest in generic expansion.
There is always a time when a patent expires, which results in a decline in the revenue of the patent owner. This time or situation caused due to patent expiration is known as a “patent cliff.” The patent cliff is the major cause of loss for an innovative pharma company as they invest a lot in their R&D and clinical trials for a particular innovation. However, it is an opportunity for the generic players in the market, which get many positive benefits from innovative companies’ recent expired patents.
Innovative Pharma Company – Four General Strategic Pathways Against Patent Cliff
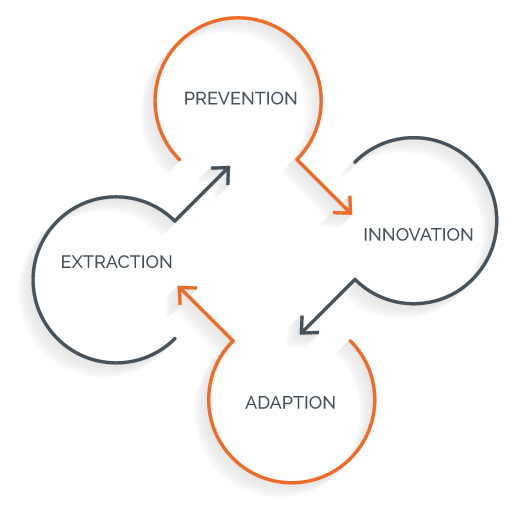
Overview of Innovative Pharma Company Strategy
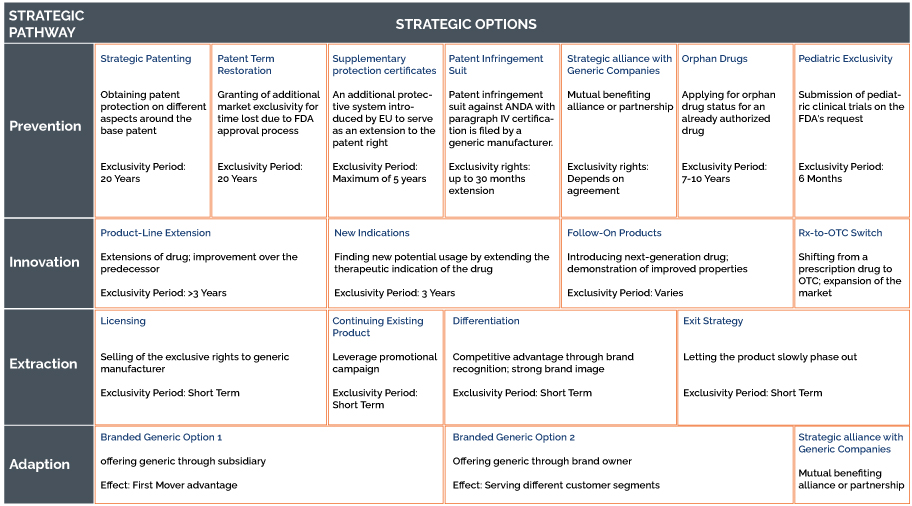
Prevention Strategy
The principle behind this strategy is to extend the patent protection and temporarily prevent or hamper the generic competition, which legal measures could make possible.
-
Strategic patenting. The pharmaceutical industry has a pattern of filling multiple patents to protect its branded drug or base patent. This pattern of filling multiple patent networks around the base patent is known as “patent clusters.” The primary patent may be split into several patents. One patent may seek protection for a broad genus encompassing various compounds, while another patent may comprise a claim related to a specific compound.
-
Another possibility to extend the market exclusivity is by pursuing a legal approach by obtaining supplementary protection certificates (SPCs) introduced by the EU to provide an extension of rights for products already on the market by a maximum of 5 years upon patent expiry.
-
In the US, the innovator companies can apply for up to 5 additional years of patent protection for the novel drug to recover the time lost while the product was subject to the FDA’s regulatory review.
-
Also, an innovator company can file a patent infringement suit after an ANDA with paragraph IV certification is filed by a generic manufacturer. The FDA cannot approve the ANDA until the court decision, thereby leading up to 30 30-month extensions of market exclusivity.
-
Another way of extending the market exclusivity is by applying for an orphan drug of the existing authorized drug, which could provide exclusivity for up to 7-10 years.
-
Generic entry can also be prevented by getting into mutual benefiting agreements with the generic manufacturer.
Innovation Strategy
The second strategy focuses on innovating the existing drug to prevent competition. In this sense, product-related innovation strategies comprise.
1. Product-line extensions
Innovator pharma companies can extend the product lifecycle by using multiple product combination methods for particular indications, which will extend the product’s life.
2. Approval of new indications
Extension by identifying novel indications for an existing drug. If the resulting evidence on possible new indications appears promising, an extension of the drug approval may be requested. Upon receiving approval for reformulation or new indications, the innovator company can acquire at least 3-years of market exclusivity.
3. Introduction of follow-on product
One can do follow-on products with follow-on products like novel drug delivery or new generation versions of the existing product. A key success factor for the utilization of this strategy is the interplay between two factors: the price sensitivity of the market and the expected degree of improvements in drug profile.
4. Rx-to-OTC-Switch
The innovator-company can either transform the prescription drug into an OTC status by weakening the dosages or solely providing the drug in OTC form. The market for OTC drugs might be a more beneficial playing ground for the brand owner to exploit the brand’s value despite the expiry of patent protection.
Extraction Strategy
Two conceptually different strategic approaches can be subsumed under this category –
-
The first strategic approach is to continue the existing product line without any further modifications to the product offering. This strategy is related to marketing campaigns or pricing strategies and cutting product-related expenses before or with the expiration of the patent to increase the company turnover.
-
The second strategic approach under this category is the selling or licensing of a patent to generic suppliers before the patent expiry. Selling or out-licensing compounds (existing patent rights and trademarks) to the generic manufacturers. An authorized generic entry is profitable if the licensing income exceeds the losses resulting from the generic entry.
Adaption Strategy
The last strategic pathway is to introduce branded generics by establishing itself as an active player in the generic market. Two conceptually similar strategic alternatives fall into this category:
- Developing a low-cost alternative in the form of a wholly-owned subsidiary focusing on generic drugs, which operates separately outside the main organization
- An offering of a generic through the innovator-company itself.
Generic Pharma Company – Three General Strategic Pathways for Generic Entry

Safe entry
The generic manufacturer has to wait for the expiry of exclusivity and patent term to launch the generic version safely. Hence, in the safe entry option, once the innovator’s drug hits the market, the launch of a generic drug generally faces two main roadblocks. Firstly, there are barriers posed by regulatory (non-patent) exclusivities, and secondly, patent and related term extensions of the innovator’s drug, which need to be surpassed for safer entry of the product.
Regulatory (Non-patent) Exclusivities
The launch of a drug in most countries is controlled by the National Drug Regulatory Authority (NDRA). Any product required to be launched must receive marketing authorization (MA) from the NDRA. The marketing authorization is granted to the company only if it can establish the drug’s quality, efficacy, and safety. The generic manufacturer is only required to establish bioequivalence to the innovator’s drug, which is highly advantageous for the generic company as it will avoid the cost of R&D and clinical trials that have already been invested by the innovator company.
Patent Exclusivity
The launch of a generic drug is prone to infringement of the innovator’s patents. A safe way of generic entry is to launch the product once the patent completes its exclusivity period (Patent expires). The ANDA applicants looking at a safe launch in the US for their generic products are required to certify their application with Paragraph 1, stating that such patent information has not been filed; Paragraph 2, certifying that such patent has expired; or Paragraph 3, certifying the date on which such patent will expire. In European countries and other countries, the requirement varies. India also has a requirement that the particulars of the patent status be furnished while filing Form 44 for seeking marketing authorization in India.
Some countries, such as the US, India, and Canada, have a provision known as a regulatory exception or Bolar exemption which gives authority to apply the patented product only after seeking marketing authorization. In India, Section 107(A)(a) of the Patent Act also provides this regulatory exemption to the generic company only after seeking proper marketing authorization.
At-Risk Entry/Launches
Not waiting for the patent to expire might seem to be a faster way of launching a generic drug. However, this strategy is associated with risks and delays stemming from patent infringement and regulatory approvals. Launching a generic drug in a market involves receiving marketing authorization from the NDRA of their respective countries. Some countries link such marketing authorizations to the patent system prevalent in their countries (patent linkage).
In countries following patent linkage systems, generic companies can get marketing authorization only if their product avoids any form of patent infringement. However, they must prove that the patented drug will not be infringed by the manufacture, use, or sale of the drug product, which will be launched by the generic company. Whereas, in countries not following the patent linkage system, the generic drug filers need to prove bioequivalence and not provide any patent-related information for the drug product.
Licensing – Voluntary and Compulsory
Another approach for a generic company to launch their product into the market is by taking licenses from innovator companies during the patent term.
Voluntary licensing is beneficial to generic companies. The innovator companies primarily decide the terms and conditions before granting the license to the other party involved.
Compulsory licensing has been provided under Article 31 of the WTO TRIPS Agreement and is complied with by all major patent systems. Under the Doha Declaration on the TRIPS Agreement and Public Health (November 2001), member nations have been given the right to grant compulsory licenses and the right to determine the grounds for granting such licenses.
Compulsory licenses are generally granted to help remedy anti-competitive practices, non-working by the patent owner, for emergency reasons, public health, or government use. The grant of a compulsory license gives the compulsory license holder exclusive rights to sell the drugs alongside the patent holder and its voluntary licensees. However, a compulsory license is less advantageous to the patent owner than a voluntary license.
Conclusion
The implementation of generic medicine was given importance in the first place to make the healthcare system in India and other countries more affordable. However, the outcomes for the innovative firm are not favorable as they have invested a lot in R&D, drug discovery, development, and clinical trials for producing a safe drug in the public interest.
The effective implementation of these strategies is yet to be explored more. The tug-of-war between the innovative and generic manufacturers will continue, as this is a never-ending cycle of discovering and exclusivity rights expiration. However, both parties will also introduce new approaches in the market for their favorable outcomes.