The advanced therapeutics medicinal products (ATMPs), particularly in the cell and gene therapies (C>) space, have grown exponentially over the last few years. New cell types and technologies have been used to overcome challenges posed by current treatments and the nature of the targeted diseases—this assists in either treating or mitigating the impact of severe disorders. The scientific and R&D efforts led to the discovery of innovative ways to engineer cells. This helps researchers address significant hurdles in complex diseases related to oncology, cardiovascular, and neurological segments. Moreover, new cell types and technologies, including CAR T Cell Therapy (Chimeric Antigen Receptor T-cell), have emerged as a transformative solution for complex oncology-related disorders.
Progress in CAR T Cell Therapy: Key Players and Development Updates
As no CAR-T cell therapy has reached clinical trial phases III/IV, all companies are striving to develop and commercialize novel antitumor treatments.
Takeda Pharma
Market giant Takeda has multiple collaborations with Noile-Immune Biotech and Miltenyi Biotec. Additionally, the acquisition of GammaDelta Therapeutics and Maverick Therapeutics has considerably strengthened its position in the R&D department. Around two years back, it also acquired Maverick’s renowned T-cell engager COBRA™ manufacturing platform.
a) R&D and Pre-clinical steps
The cellular composition and immunophenotype of the administered cells play a crucial role in therapeutic success.
b) Clinical developments: Phase I and Phase II
- TAK-981 is being tested to evaluate safety, tolerability, preliminary efficacy, and pharmacokinetics (PK) in advanced or metastatic solid tumors or relapsed/refractory hematologic malignancies.
- The study in Phase 2 is focused on dose expansion cohorts (cancer treatment expansions).
c) Regulatory information
No drug has reached clinical trial phase III/IV, thus no FDA approval is granted.
Bellicum
The midsize pharmaceutical company has been actively developing treatments for multiple cancers, such as pancreatic and HER 2+ cancers.
a) R&D and Pre-clinical steps
GoCART – The company’s proprietary CAR-T cell therapy platform to target solid tumors.
b) Clinical developments: Phase I and Phase II
- Phase 1: Cell dose escalation studies to identify the maximum dose of BPX-601 administered with single or repeat doses of Rimiducid.
- Phase 2: Indication-specific dose expansion studies to assess the safety and pharmacodynamics (including BPX-601 persistence)
c) Regulatory information
No drug has reached clinical trial phase III/IV, thus, no FDA approval granted.
Person Gen Therapeutics
The startup, founded in 2010, has registered 13 CAR-T and CAR-NK clinical trials so far. The company offers diverse platforms and a vibrant pipeline targeting multiple solid tumors.
a) R&D and Pre-clinical steps
The company has multiple platforms centered around CAR.
- SAFEY UCAR PLATFORM
- CAR Structure Functional Verification Platform
- Automated CAR T manufacturing platform
- In vivo CAR T efficacy verification platform
b) Clinical developments: Phase I and Phase II
- Anti-MUC1-CAR T CELLS against gastric carcinoma, colorectal carcinoma, and malignant glioma.
- Another Trial Phase I/II Study of Anti-Mucin1 (MUC1) CAR T Cells for Patients With MUC1+ Advanced Refractory Solid Tumor
c) Regulatory information
No drug has reached clinical trial phase III/IV, thus no FDA approval granted.
Recent Advancements in CAR T Cell Therapy for Solid Tumors
Numerous enterprises have achieved significant progress around CAR-T cell therapy development by aiming to overcome challenges like tumor heterogeneity and the immunosuppressive tumor microenvironment. A few of the key developments by recognized organizations that strive to take cancer treatment to the next level are:
Syn-Notch CAR Circuits
CAR-T cells have encountered roadblocks in solid tumors, including the lack of highly tumor-specific antigens to target. This elevates the possibility of life-threatening ‘on-target/off-tumor’ toxicities. Additionally, there are problems with T cell entry into the solid tumor and persistent activity in suppressive tumor microenvironments.
SynNotch (Synthetic Notch) CAR-T cells display superior control of tumor burden compared to T cells. These engineered cells constitutively express a CAR targeting the same antigens in mouse models of human mesothelioma and ovarian cancer.
Exhibit: synNotch Circuit
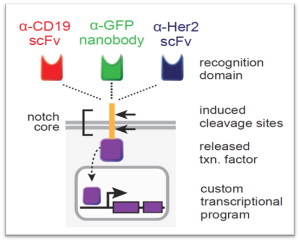
Researchers at the University of California achieved this by preventing CAR-mediated tonic signaling through synNotch-controlled expression, allowing T cells to maintain a long-lived memory and non-exhausted phenotype. Collectively, they have established ALPPL2 as a clinically viable cell therapy target for multiple solid tumors and demonstrated the multifaceted therapeutic benefits of synNotch CAR-T cells.
Anixa’s Chimeric Endocrine Receptor (CER)-T approach for Ovarian Cancers
Anixa’s novel CAR-T approach, known as “Follicle Stimulating Hormone Receptor (FSHR)-mediated CAR-T technology, recently got its patent approved in China. In CER-T, engineered T-cells target the follicle-stimulating hormone receptor (FSHR) in patients with Ovarian Epithelial cancer.
Exhibit: Anixa’s CER-T Approach
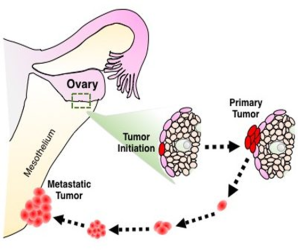
Design of CARs: The team designed the CAR constructs by incorporating the signal peptide of murine CD8a, which was followed by a fusion of full-length murine FSHb and CGa peptides, connected by a glycine/serine spacer. The construct also included the murine CD8a hinge, transmembrane domain, and an intracellular fragment of murine 4-1BB and CD3z.
Rationale of the Study: The follicle-stimulating hormone (FSH) receptor is expressed on the cell surface of ovarian carcinomas of different histologic types, as well as on clear cell and mucinous tumors, which are particularly aggressive. Most importantly, no FSHR expression is found in nonovarian healthy tissues in adult women. Therefore, T cells redirected against FSHRþ tumor cells with full-length FSH represented a promising therapeutic alternative against a broad range of ovarian malignancies. This approach has negligible toxicity, even in the presence of cognate targets in tumor-free ovaries.
Nanobody-based CARs
Approved CARs are based on monoclonal antibodies or SCFV (single-chain fragment variable) for target identification. SCFV-based CARs have several problems, ranging from losing their binding affinity post conversion from IGG to difficulties finding the most optimal linker and V-region orientation. The predominant usage of mouse-based SCFVs elicits the anti-immune response, with a tendency to aggregate along with their non-specific interactions.
Exhibit: Nanobody-based CARs
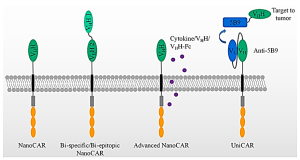
CAR nanobodies serve as the antigen-binding domain of CAR-T owing to their small size, optimal stability, high affinity, and manufacturing feasibility.
Many companies, such as Bluebird Bio, Autolus, and the University of Pennsylvania, have actively developed Nanobody-based CAR constructs targeting solid tumors.
LEGEND BIOTECH’s (NCT03090659) clinical trial for multiple myeloma is currently in phase 2. Their construct comprises VHH incorporated into CAR.
Exemplary collaborations and partnerships in the last three years
MUSC Hollings Cancer Center + Lowvelo


Researchers’ project, started at MUSC Hollings Cancer Center, focuses on studies that reduce the side effects associated with CAR-T-cell therapy and provide better treatment options for patients. LOWVELO’s grant supported the project’s funding.
Chimeric Therapeutics + WUXI ATC


Chimeric Therapeutics will leverage WuXi ATU’s end-to-end closed-loop CAR-T process development, Good Manufacturing Practice (GMP) manufacturing, and testing platform to develop its drug CHM 2101.
Transgene + PERSON GEN THERAPEUTICS


The strategic collaboration aims to evaluate the feasibility and efficacy of combination therapy. This therapy associates PersonGen’s TAA06 CAR-T cell injection with intravenous (IV) administration of an armed oncolytic virus from Transgene’s Invir.IO™ platform for solid tumors, including pancreatic and brain glioma.
Conclusion
At Ingenious e-Brain (IeB), we stand at the forefront of technological advancement, offering invaluable technological scouting and benchmarking services that enable clients to select optimal platforms for scaling up their endeavors. Moreover, as part of IeB’s exclusive IP solutions spectrum, we extend our support by identifying available licensing opportunities, empowering clients with a keen understanding of the current landscape and potential collaborations in cancer care. We offer tailored solutions finely tuned to meet each client’s unique needs, ensuring a strategic advantage in navigating the intricate terrain of innovation and discovery.
Want to talk to our seasoned IP experts in the life science domain? Either fill out the form below or email them to contact@iebrain.com.